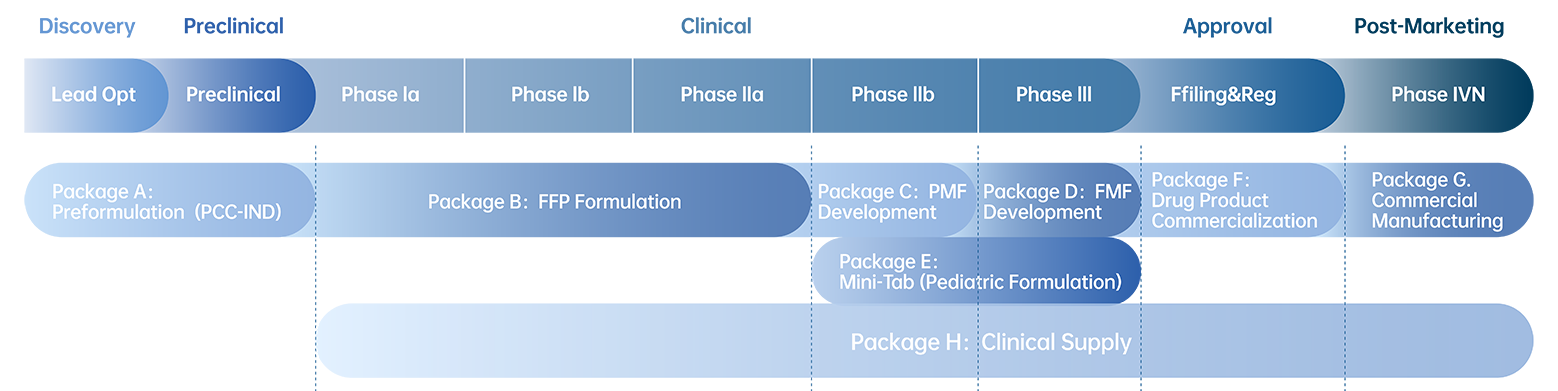
◆ Preformulation and material characterization
◆ Formulation development and manufacturing service from preclinical, clinical to commercialization (including Spray Drying and Holt Melt Extrusion)
◆ Integrated one-stop shop of clinical drugs
◆ cGMP and QMS system meets NMPA, FDA, and EMA requirement
◆ CMC filing support for IND/CTA/CSA in China and overseas




Preformulation
A systematic preformulation study is an important preparation for formulation development. It provides the basis...
Formulation Development
Crystal Formulation Service boasts a formulation development leadership team with extensive experience and deep expertise...
Clinical and Commercial Manufacturing Service
We have the following capabilities,an experienced team with high efficient execution,world-class manufacturing equipment...
Analytical Chemistry
We will provide our valued customers with analytical chemistry research and quality control services for NCEs throughout ...
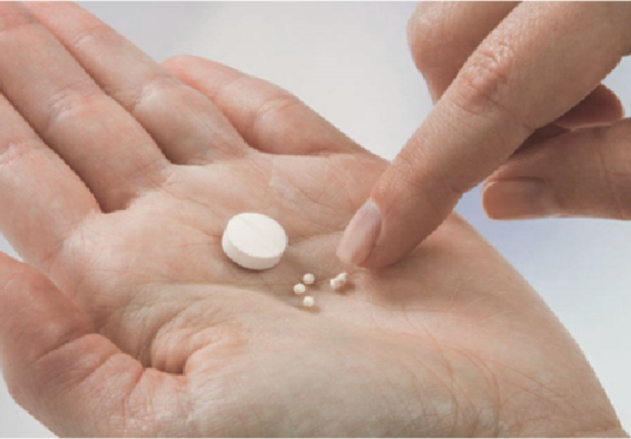


Dosage forms designed for pediatric use has long been an unmet medical need. The lack of pediatric formulation has led to ...
more >As the demand for small molecule drugs with high bioavailability continues to grow, there is an increased demand for...
more >Most small molecule drugs are formulated as oral solid dosage (OSD) forms, such as tablets or capsules.
more >